Desmognathus
Allan Larson, David Wake, and Tom Devitt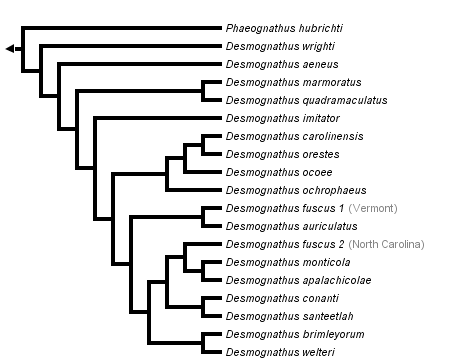


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
Salamanders of the plethodontine supergenus Desmognathus are widely distributed in the forests of eastern North America. Three species (Desmognathus aeneus, Desmognathus wrighti and Phaeognathus hubrichti) are strictly terrestrial and lack an aquatic larval stage. Eggs are laid on land and hatch fully formed. All remaining species belong to the genus Desmognathus and have biphasic life histories featuring gilled, aquatic larvae and fully metamorphosed adults.
Ecological communities in the southern Appalachian Mountains commonly feature three to five species of Desmognathus and may have as many as seven Desmognathus species (Bruce, 1991; Tilley and Bernardo, 1993). These communities figure prominently in experimental ecological studies (reviewed by Hairston, 1987). The different species of Desmognathus present in these communities demonstrate considerable differentiation in the degree to which they use aquatic versus terrestrial environments. Desmognathus marmoratus is entirely aquatic in both the larval and adult stages and inhabits cold, fast-flowing streams. It has a long larval period (almost 3 years) and a relatively large body size, which may exceed 14 cm. in total length. A related species, D. quadramaculatus, also has a large body (may exceed 20 cm. total length), long larval period (3-4 years) and is mostly aquatic as an adult, favoring swift mountain streams. Intermediate-sized species of Desmognathus (total body length approaching a maximum of 10-17 cm. in different species) inhabit edges of streams and seepages, and have larval periods of intermediate length (~4 months to a year); these species include D. carolinensis, D. conanti, D. fuscus, D. imitator, D. monticola, D. ochrophaeus, D. ocoee, D. orestes, D. santeetlah, and D. welteri. The directly developing, entirely terrestrial Desmognathus species (D. aeneus, D. wrighti) have the smallest bodies in the supergenus Desmognathus (less than 6 cm maximum total length).
Traditionally, the semiaquatic species D. quadramaculatus was considered to approximate the ancestral condition for Desmognathus with the other states representing evolutionarily derived states; however, the phylogenetic results of Titus and Larson (1996) challenge this interpretation by showing that the terrestrial species span the earliest phylogenetic splits in the subfamily, as illustrated above. Titus and Larson (1996) suggest that desmognathine evolution features primarily increases in body size, length of larval period and use of aquatic habitats as adults.
The genus Desmognathus has been the source of some very interesting studies of the evolution of sexual isolation among populations and its relationship to population genetic differentiation (Tilley et al., 1990; Verrell, 1989, 1994; Verrell and Arnold, 1989; Verrell and Tilley, 1992). These studies are particularly important for our understanding of processes of speciation in salamanders.
Characteristics
Diagnosis
Salamanders of the supergenus Desmognathus are woodland salamanders whose habitats vary from strictly terrestrial to strictly aquatic. Members of this supergenus are characterized by a number of characters that are associated functionally with burrowing under rocks and include (1) a heavily ossified and strongly articulated skull and mandible, (2) a dorsoventrally flattened, wedgelike head, (3) modified anterior trunk vertebrae, (4) enlarged dorsal spinal muscles and (5) hindlimbs larger than forelimbs. Three additional characters appear functionally associated with feeding and include (1) stalked occipital condyles, (2) a modified atlas and (3) enlarged quadratopectoralis muscles. A ninth character, presence of atlanto-mandibular ligaments, appears to be important for both burrowing and feeding (Schwenk and Wake, 1993).
Detailed Characteristics of the Supergenus Desmognathus
In addition to the nine characteristics listed above under "Diagnosis," some additional characteristics are useful in combination for distinguishing desmognathine salamanders from other plethodontids; however, most of these characteristics do not constitute synapomorphies of the supergenus Desmognathus. These characteristics are summarized from Lombard and Wake's (1986) phylogenetic analysis of major plethodontid lineages with special emphasis on evolution of feeding mechanisms.
Tongue and Hyobranchial Apparatus
Tongues are attached to the jaw by a short muscle plus connective and mucosal tissue. Tongues are protrusible but not projectile. The hyobranchial skeleton includes a urohyal, cylindrical basibranchial, and radii that are rodlike structures independent of the basibranchial. The basibranchial, first ceratobranchial and epibranchial elements are of approximately equal length. The first ceratobranchial is larger than the second ceratobranchial and constitutes the main force-transmitting element in movement of the tongue. The rectus cervicis profundis muscle is linearly arranged and the rectus cervicis superficialis has a lateral slip. Omohydoideus, genioglossus, circumglossus, intraglossus and basiradialis muscles are present. The hyoglossus muscle is large and has a complete anterior section and posteriorly oriented fibers in the posterior section. The suprapeduncularis muscle is weakly developed.
Epibranchial Number
Embryos (or larvae) have four epibranchials.
Tail Autotomy
Cutaneous wound healing does not occur, there are 2 caudosacral vertebrae, the first caudal vertebra is normal, and tail breakage is not localized.
Brain Stem Motor Column
There are two distinct classes of cells in the motor column of the neck and trunk.
Jaws, Cranial Osteology and Structure of the Inner Ear
The vomer has a bony shelf anterior to the preorbital process. Parietal bones have a deep groove extending across the posterolateral portion of the bone to accommodate the ligament that extends from the lower jaw to the atlas. The facial process of the maxilla is large and extends into the area vacated by the prefrontal bones. The premaxillae are fused and surround the intermaxillary gland. In the inner ear, the amphibian periotic canal forms a ventral loop immediately after leaving the periotic cistern. The bore radius of the otic semiarticular ducts has a strong negative allometry with respect to body weight.
Chromosome Number
The diploid number of chromosomes is 28.
Development
All but 3 species (Desmognathus aeneus, D. wrighti and Phaeognathus hubrichti) have gilled, aquatic larvae.
Classification
The supergenus Desmognathus contains only two genera, Desmognathus (17 species currently recognized; Tilley and Mahoney, 1996; Titus and Larson, 1996) and the monotypic genus Phaeognathus. Desmognathus marmoratus formerly was placed into the monotypic genus, Leurognathus. Recognition of Leurognathus was discontinued by Titus and Larson (1996) because such recognition clearly makes Desmognathus paraphyletic.
Discussion of Phylogenetic Relationships
The strongest conclusions regarding desmognathine phylogeny that can be made from the analysis of Titus and Larson (1996) are (1) monophyly of Desmognathus (including D. marmoratus), (2) Desmognathus and Phaeognathus form sister taxa, (3) D. wrighti is the sister taxon to all other Desmognathus combined, (4) D. marmoratus and D. quadramaculatus together form a monophyletic group, (5) D. conanti and D. santeetlah are sister species, and (6) D. auriculatus is closely related to northern populations of D. fuscus. Uncertainties include the exact phylogenetic position of D. aeneus, although the most parsimonious tree makes it the sister taxon to all metamorphosing Desmognathus. Monophyly of D. fuscus is uncertain even after D. conanti, formerly a subspecies of D. fuscus, is treated as a separate species. Exact relationships among lineages of metamorphosing Desmognathus, except for those listed above, are very tentative, including relationships among members of the D. ochrophaeus complex (Tilley and Mahoney, 1996).
References
Bruce, R. C. 1991. Evolution of ecological diversification in desmognathine salamanders. Herpetological Review 22:44-46.
Chippindale, P. T., R. M. Bonett, A. S. Baldwin, and J. J. Wiens. 2004. Phylogenetic evidence for a major reversal of life-history evolution in plethodontid salamanders. Evolution 58:2809-2822.
Hairston, N. G. 1987. Community Ecology and Salamander Guilds. Cambridge Univ. Press. Cambridge, England.
Larson, A. 1984. Neontological inferences of evolutionary pattern and process in the salamander family Plethodontidae. Evolutionary Biology 17:119-217.
Lombard, R. E. and D. B. Wake. 1986. Tongue evolution in the lungless salamanders, family Plethodontidae. IV. Phylogeny of plethodontid salamanders and the evolution of feeding dynamics. Systematic Zoology 35:532-551.
Schwenk, K. and D. B. Wake. 1993. Prey processing in Leurognathus marmoratus and the evolution of form and function in desmognathine salamanders. Biological Journal of the Linnean Society 49:141-162.
Tilley, S. G. and J. Bernardo. 1993. Life history evolution in plethodontid salamanders. Herpetologica 49:154-163.
Tilley, S. G. and M. J. Mahoney. 1996. Patterns of genetic differentiation in salamanders of the Desmognathus ochrophaeus complex (Amphibia: Plethodontidae). Herpetological Monographs 10:1-42.
Tilley, S. G., P. A. Verrell and S. J. Arnold. 1990. Correspondence between sexual isolation and allozyme differentiation: A test in the salamander Desmognathus ochrophaeus. Proceedings of the National Academy of Sciences, USA 87:2715-2719.
Titus, T. A. and A. Larson. 1996. Molecular phylogenetics of desmognathine salamanders (Caudata: Plethodontidae): A reevaluation of evolution in ecology, life history, and morphology. Systematic Biology 45:451-472.
Verrell, P. A. 1989. An experimental study of the behavioral basis of sexual isolation between two sympatric plethodontid salamanders, Desmognathus imitator and D. ochrophaeus. Ethology 80:274-282.
Verrell, P. A. 1994. Evidence against a role for experience in the maintenance of sexual incompatibility between sympatric salamanders. Herpetologica 50:475-479.
Verrell, P. A. and S. J. Arnold. 1989. Behavioral observations of sexual isolation among allopatric populations of the mountain dusky salamander, Desmognathus ochrophaeus. Evolution 43:745-755.
Verrell, P. A. and S. G. Tilley. 1992. Population differentiation in plethodontid salamanders: Divergence of allozymes and sexual compatibility among populations of Desmognathus imitator and Desmognathus ochrophaeus (Caudata: Plethodontidae). Zoological Journal of the Linnean Society 104:67-80.
Wake, D. B. 1966. Comparative osteology and evolution of the lungless salamanders, family Plethodontidae. Memoirs of the Southern California Academy of Sciences 4:1-111.
Title Illustrations

| Scientific Name | Desmognathus fuscus |
|---|---|
| Location | New York |
| Comments | with eggs |
| Specimen Condition | Live Specimen |
| Copyright | © 1996 Greg Sievert |
| Scientific Name | Phaeognathus hubrichti |
|---|---|
| Location | Alabama |
| Copyright | © 1996 Greg Sievert |
About This Page
David Heyse, Todd Jackman and Greg Sievert contributed to the preparation of this Tree of Life page.
Allan Larson

Washington University, St. Louis, Missouri, USA
David Wake

University of California, Berkeley, California, USA
Tom Devitt

University of California, Berkeley, California, USA
Correspondence regarding this page should be directed to David Wake at
Page copyright © 2006 Allan Larson, David Wake, and
All Rights Reserved.
- Content changed 26 September 2006
Citing this page:
Larson, Allan, David Wake, and Tom Devitt. 2006. Desmognathus. Version 26 September 2006 (under construction). http://tolweb.org/Desmognathus/15529/2006.09.26 in The Tree of Life Web Project, http://tolweb.org/









 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site