raphid diatoms
David G. Mann


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
This is by far the largest group of diatoms in terms of numbers of species. It is a natural group, easily recognized by the presence of a unique organelle, the raphe system, which is involved in locomotion: cells use it to glide over surfaces and through sediments. No other group of unicellular autotrophs (plants) has evolved such an effective method of coping with life in mobile muds and sands. Average speeds of around 5-20 µm per second are commonly achieved in the laboratory, which means that a diatom (perhaps 50 µm long) could potentially travel several centimetres in an hour. It is unlikely that this ever happens, because reversals of direction are frequent, but raphid diatoms are certainly able to migrate significant distances up and down in sediments, and can keep pace with quite rapid burial by fresh sediment. They also use their motility to find mates during sexual reproduction.
Characteristics
- usually highly motile, through the activity of a unique organelle, the raphe system, consisting of one or two slits through the valve (but lost through secondary infilling in a few cases) and secretory and cytoskeletal components within the protoplast.
- raphe slits either closely associated with the sternum or, more commonly, fully integrated into it and forming a compound pattern-centre, the 'raphe-sternum'.
- formation of the two sides of the valve asynchronous and dissimilar: consequently, primary and secondary sides can be distinguished.
- plastids usually few in number and large, often lobed and so positioned in the cell as to avoid the regions beneath the raphe systems.
- sexual reproduction achieved through active pairing of sexualized vegetative cells; secondary reduction or loss of sexuality occurs but is uncommon, leading to auto- or apomictic auxosporulation.
- auxospore expansion bipolar, usually constrained by a perizonium consisting of transverse and longitudinal bands, as in some araphid pennate diatoms (e.g. Rhabdonema).
Discussion of Phylogenetic Relationships
Old classifications of diatoms (Karsten 1928, Hustedt 1927-66) split the raphid diatoms into three suborders, namely:
- Raphidioideae (Eunotia, Peronia, Actinella)
- Monoraphideae (Achnanthes, Campyloneis, Cocconeis)
- Biraphideae (all other raphid diatoms
These three, together with the suborder Araphideae (raphe-less or araphid pennate diatoms), comprised the Pennales (pennate diatoms). The Raphidioideae were characterized by having a rudimentary raphe system ("Raphenanfang vorhanden": Karsten 1928), the Monoraphideae had a true raphe on one valve in each frustule, and the Biraphideae had a true raphe on both valves of each frustule.
Ultrastructural, cytological and molecular data seem to confirm that the Raphidioideae, which are now called either Eunotiophycidae or Eunotiales (there is no consensus on what ranks should be used), are an isolated, monophyletic group. The balance of evidence suggests that they are sister to a monophyletic group containing all other raphid diatoms, but support for this from molecular data is, so far, equivocal (e.g. Alverson et al. 2006, Medlin & Kaczmarska 2004, Sorhannus 2007).
One feature that suggests a basal dichotomy between the Eunotiophycidae and other raphid diatoms is that they possess the special lipped structures called rimoportulae (= rimoportules, labiate processes), which are found in most clade I and clade II araphid pennate diatoms and in most centric diatom lineages, but in no other raphid diatoms apart from the Eunotiophycidae. Thus, if the Eunotiophycidae are not a basal group but branch off further within the raphid diatoms (e.g. as suggested by the SSU rDNA tree of Sorhannus 2007), then rimoportulae must have been lost several or many times in the raphid group; there is no evidence for this, e.g. from the fossil record. Also, the raphe system of Eunotiophycidae may or may not be 'rudimentary', as suggested by Karsten, but it is certainly quite differently positioned relative to the sternum and striae than in all other raphid diatoms and appears to represent a less complete stage in the integration of the raphe into the pattern-centre.
The Monoraphideae are not a monophyletic group. Heterovalvy, in which one valve of each frustule possesses a raphe and the other does not, has evolved at least twice, in Achnanthes sensu stricto, and in the Cocconeis–Achnanthidium clade (e.g. Sorhannus 2007, fig. 2: Achnanthes longipes is mislabelled as 'Achnanthidium longipes', following an incorrect older nomenclature); these are just two major clades among many within the Biraphideae, here referred to as Bacillariophycidae.
References
Alverson, A.J., Cannone, J.J., Gutell, R.R. & Theriot, E.C. (2006). The evolution of elongate shape in diatoms. Journal of Phycology 42: 655-668.
Edgar, L.A. & Pickett-Heaps, J.D. (1984). Diatom locomotion. Progress in Phycological research 3: 47-88.
Hustedt, F. (192766). Die Kieselalgen Deutschlands, Österreichs und der Schweiz unter Berücksichtigung der übrigen Länder Europas sowie der angrenzenden Meeresgebiete. In Dr. L. Rabenhorsts Kryptogamenflora von Deutschland, Österreich und der Schweiz, vol. 7. Akademische Verlagsgesellschaft, Leipzig.
Karsten,G. (1928). Bacillariophyta (Diatomaceae). In: Die Natürlichen Pflanzenfamilien, 2nd ed. (Ed. A. Engler & K. Prantl), vol. 2 Leipzig, W. Engelmann, pp. 105203.
Mann, D.G. (1996). Chloroplast morphology, movements and inheritance in diatoms. In B.R. Chaudhary & S.B. Agrawal (eds) Cytology, genetics and molecular biology of algae, pp. 249-274. SPB Academic Publishing, Amsterdam.
Medlin, L.K. & Kaczmarska, I. (2004). Evolution of the diatoms: V. Morphological and cytological support for the major clades and a taxonomic revision. Phycologia 43: 245270.
Palmer, J.D. & Round, F.E. (1967). Persistent, vertical-migration rhythms in benthic microflora. VI. The tidal and diurnal nature of the rhythm in the diatom Hantzschia virgata. Biological Bulletin 132: 44-55.
Round, F.E., Crawford, R.M. & Mann, D.G. (1990). The diatoms. Biology and morphology of the genera. Cambridge University Press, Cambridge. 747 pp.
Sorhannus, U. (2007). A nuclear-encoded small-subunit ribosomal RNA timescale for diatom evolution. Marine Micropaleontology 65: 1-12.
Title Illustrations

| Scientific Name | Lyrella hennedyi |
|---|---|
| Comments | Optical focus near one valve; two complex lobed chloroplasts are present, one towards each pole of the cell. |
| Specimen Condition | Live Specimen |
| Identified By | David Mann |
| Life Cycle Stage | Vegetative phase |
| Body Part | Single cell |
| View | Valve view |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© David G. Mann

|
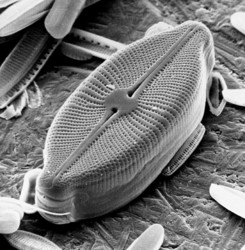
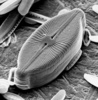
| Scientific Name | Cosmioneis pusilla |
|---|---|
| Location | underside of mediaeval bridge, Llanymddyfri, Dyfed, Wales |
| Comments | Scanning electron micrograph |
| Specimen Condition | Dead Specimen |
| Identified By | David Mann |
| Life Cycle Stage | Vegetative phase |
| Body Part | Frustule |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 2008 David G. Mann

|
| Scientific Name | Neidium |
|---|---|
| Location | Ashford in the Water, Derbyshire, England |
| Specimen Condition | Live Specimen |
| Identified By | David Mann |
| Life Cycle Stage | Vegetative phase |
| Body Part | Whole cell |
| View | Median optical section |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 2008 David G. Mann

|
About This Page
This page is being developed as part of the Tree of Life Web Project Protist Diversity Workshop, co-sponsored by the Canadian Institute for Advanced Research (CIFAR) program in Integrated Microbial Biodiversity and the Tula Foundation.
David G. Mann

Royal Botanic Garden Edinburgh, United Kingdom
Correspondence regarding this page should be directed to David G. Mann at
Page copyright © 2008 David G. Mann
 Page: Tree of Life
raphid diatoms.
Authored by
David G. Mann.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
raphid diatoms.
Authored by
David G. Mann.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
- First online 07 February 2010
Citing this page:
Mann, David G. 2010. raphid diatoms. Version 07 February 2010 (under construction). http://tolweb.org/raphid_diatoms/125307/2010.02.07 in The Tree of Life Web Project, http://tolweb.org/









 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site