Evanioidea
Andrew R. Deans and John T. Jennings

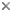
This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
Evanioidea comprises three families, Aulacidae, Gasteruptiidae, and Evaniidae. Systematists have consistently uncovered morphological (Naumann 1991, Whitfield et al. 1989, Gauld and Bolton 1988, Gibson 1985, Carlson 1979, Crosskey 1951), biological (Gauld and Bolton 1988, Crosskey 1951, Bradley 1908), and molecular (Dowton and Austin 2001, Dowton et al. 1997) evidence questioning the monophyly of Evanioidea, but no formal higher level taxonomic alterations have been made since Hedicke (1939).
Characteristics
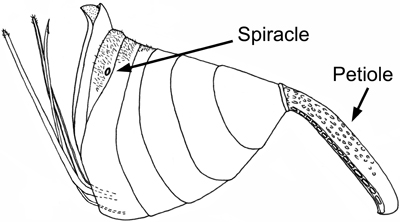
Evanioidea share two possible apomorphies: the dorsal articulation of the metasoma to the mesosoma (Fig. 1) and the loss of all functional metasomal spiracles (breathing holes) except on the seventh segment (Figs. 1 and 2).
Figure 1. Diagram of a female Evanioid (Gasteruptiidae) showing the dorsal articulation of the metasoma to the mesosoma (after Goulet and Huber 1993).
Figure 2. Evanioid (Evaniidae) metasoma (female) showing reduction of spiracles except for on the seventh segment. Image copyright ? 2004 Andrew R. Deans.
Are Evanioidea monophyletic?
The most apparent evidence suggesting a non-monophyletic Evanioidea are the differences in host biology for the three families. Evaniids oviposit into cockroach oothecae buried in substrate, loose in leaf litter, or attached to female cockroaches. Gasteruptiids are predator-inquilines that lay eggs inside the cells of solitary bees and wasps nesting in plant stems or in underground nests, with the subsequent larvae developing on the food stores and/or nest inhabitants (Jennings and Austin 2004). Aulacids, considered by some hymenopterists to be the most ancestral of the three families, are endoparasitic on wood-boring xiphydriid sawflies and cerambycid and buprestid beetles (Jennings and Austin 2003). Many hymenopterists hypothesize an easy transition from parasitizing wood-boring larval hosts to "parasitizing" nest cells within plant stems but not to "parasitizing" cockroach egg cases (Whitfield 1998). Gasteruptiidae and Aulacidae also share more synapomorphies with each other than with Evaniidae and have frequently been combined into one family (Gasteruptiidae; see Townes 1950). These facts, combined with evidence that species in several other distantly related lineages (Chalcidoidea, Cynipoidea, Ichneumonoidea) independently evolved dorsally articulated metasomas, have led people to question the monophyly of Evanioidea.
Discussion of Phylogenetic Relationships
Despite conflicting evidence several recent phylogenetic and other studies predict an Evaniidae + Gasteruptiidae clade. Ronquist et al. (1999), using morphological data, recovered these two families as sister taxa with low support. Quicke et al. (1994) found some similarities between the ovipositor in Aulacidae and Gasteruptiidae, but not Evaniidae. Both families have a medial thickening of the ventral wall of the upper valve, but the Gasteruptiidae have a mid-dorsal longitudinal ridge that is absent in aulacids. Evaniidae differ in that their ovipositor is dorso-ventrally compressed rather than diverging, a character that may be functionally linked to parasitising blattodean oothecae.
Deans et al. (2004) and Mason (1993) noted that several amber fossils (Fig. 3) reveal intermediate forms between Aulacidae and Evaniidae, and Jennings et al. (2004) noted that Hyptiogastrites electrinus Cockerell from Burmese amber (Fig. 4) exhibits characteristics of both Gasteruptiidae and Aulacidae. Dowton and Austin (2001) also recovered Evaniidae + Gasteruptiidae using evidence from three genes (Fig. 5 in Dowton and Austin 2001) and three genes plus morphological characters (Figs. 7,9,13 in Dowton and Austin 2001). However, their placement of Evaniidae depended on the tree building methods and models employed. Sister-group relationships also resulted between ensign wasps (Evaniidae) and other taxa, mainly Evaniidae + Heloridae (Figs. 2,3,6 in Dowton and Austin 2001) and Evaniidae + Ceraphronoidea (Figs. 4,10 in Dowton and Austin 2001). Unpublished reanalyses using Dowton and Austin's data (Deans and Whitfield 2003, using Bayesian methods) recovered a monophyletic Evanioidea 100% of the time, but the relationships between families differed depending on the assumptions made.
Figure 3. Evaniid fossil in Lebanese amber with some characteristics of the family Aulacidae. Image copyright © 2004 Andrew R. Deans.
Figure 4. Hyptiogastrites electrinus Cockerell in Burmese amber exhibiting characteristics of both Aulacidae and Gasteruptiidae. Image copyright © 2004 John T. Jennings.
References
Bradley, J. C. 1908. The Evaniidae, ensign flies, an archaic family of Hymenoptera. Transactions of the American Entomological Society 34: 101-194.
Carlson, R. W. 1979. Superfamily Evanioidea. Pp. 1109-1118 in: Catalog of the Hymenoptera in America North of Mexico, volume 1, Symphyta and Apocrita (Parasitica). K. V. Krombein, P. D. Hurd, D. R. Smith, and B. D. Burks, eds. Smithsonian Institution Press, Washington, D. C., U.S.A. 1198 pp.
Crosskey, R. W. 1951. The morphology, taxonomy, and biology of the British Evanioidea (Hymenoptera). Transactions of the Royal Entomological Society of London 102: 247-281.
Deans, A. R., H. H. Basibuyuk, D. Azar, and A. Nel. 2004. Descriptions of two new Early Cretaceous (Hauterivian) ensign wasp genera (Hymenoptera: Evaniidae) from Lebanese amber. Cretaceous Research 25: 509-516.
Deans, A. R. and J. B. Whitfield. 2003. Estimates of relationships between ensign wasp genera (Hymenoptera: Evaniidae). Evolution Conference 2004. Fort Collins, CO. June 26-30, 2004.
Dowton, M. and A. D. Austin. 2001. Simultaneous analysis of 16S, 28S, COI and morphology in the Hymenoptera: Apocrita - evolutionary transitions among parasitic wasps. Biological Journal of the Linnaean Society 74: 87-111.
Dowton, M., A.D. Austin, N. Dillon, and E. Bartowsky. 1997. Molecular phylogeny of the apocritan wasps with particular reference to the Proctotrupomorpha and Evaniomorpha. Systematic Entomology 22: 245-255.
Gauld, I. D. and B. Bolton. 1988. The Hymenoptera. Oxford University Press, Oxford, U.K. 332 pp.
Gibson, G. A. P. 1985. Some pro- and mesothoracic structures important for phylogenetic analysis of Hymenoptera, with a review of the terms used for the structures. Canadian Entomologist 117: 1395-1443.
Goulet, H. and J. T. Huber (eds.) 1993. Hymenoptera of the World: An Identification Guide to Families. Research Branch, Agriculture Canada Publication 1894/E. 668 pp.
Hedicke, H. 1939. Evaniidae. In: Hymenoptorum Catalogus, H. Hedicke (ed.) Pars 9. Dr. W. Junk. 's-Gravenhage, 50 pp.
Jennings, J. T. and A. D. Austin. 2004. Biology and host relationships of aulacid and gasteruptiid wasps (Hymenoptera: Aulacidae): a review. Pp. 187-215. In K. Rajmohana, K. Sudheer, P. Girish Kumar, and S. Santhosh (eds.) Perspectives on Biosystematics and Biodiversity. University of Calicut, Kerala, India.
Jennings, J. T., A. D. Austin, and N. B. Stevens. 2004. Hyptiogastrites electrinus Cockerell, 1917, from Burmese amber: redescription and its placement within the Evanioidea (Hymenoptera). Journal of Systematic Palaeontology 2: 127-132.
Mason, W. R. M. 1993. Superfamilies Evanioidea, Stephanoidea, Megalyroidea, and Trigonalyoidea. Chapter 11, pp. 510-520 In: Goulet, H. and J. T. Huber, eds. Hymenoptera of the World: An identification guide to families. Research Branch, Agriculture Canada, Publication 1894/E. 668 pp.
Naumann, I. D. 1991. Hymenoptera. Pp. 916-1000 In: The Insects of Australia, Volume II. Melbourne University Press, Melbourne, Australia.
Quicke, D. L. J., M. G. Fitton, J. R. Tunstead, S. N. Ingram, and P. V. Gaitens. 1994. Ovipositor structure and relationships within the Hymenoptera, with special reference to the Ichneumonoidea. Journal of Natural History 28: 635-682.
Ronquist, F., A. P. Rasnitsyn, A. Roy, K. Eriksson, and M. Lindgren. 1999. Phylogeny of the Hymenoptera: a cladistic reanalysis of Rasnitsyn's (1988) data. Zoologica Scripta 28: 13-50.
Sharkey, M. J. and A. Roy. 2002. Phylogeny of the Hymenoptera: a reanalysis of the Ronquist et al. (1999) reranalysis, emphasizing wing venation and apocritan relationships. Zoologica Scripta 31: 57-66.
Townes, H. K. 1950. The Nearctic species of Gasteruptiidae (Hymenoptera). Proceedings of the United States National Museum 100: 85-145.
Whitfield, J. B. 1998. Phylogeny and evolution of host-parasitoid interactions in Hymenoptera. Annual Review of Entomology 43: 129-151.
Whitfield, J. B., N. F. Johnson, and M. R. Hamerski. 1989. Identity and phylogenetic significance of the metapostnotum in nonaculeate Hymenoptera. Annals of the Entomological Society of America 82: 663-673.
Title Illustrations
| Scientific Name | Aulacidae |
|---|---|
| Location | Australia |
| Specimen Condition | Dead Specimen |
| Sex | Female |
| Life Cycle Stage | adult |
| Body Part | habitus |
| Size | ~12mm |
| Copyright | © 2004 John T. Jennings |
| Scientific Name | Evaniidae |
|---|---|
| Location | Honduras |
| Creator | Andrew. R. Deans |
| Specimen Condition | Dead Specimen |
| Sex | Female |
| Life Cycle Stage | adult |
| Body Part | habitus |
| Size | ~11mm |
| Copyright | © 2004 Andrew R. Deans |
About This Page
Andrew R. Deans
School of Computational Science
Florida State University
Tallahassee, FL 32306
U.S.A.
John T. Jennings
Waite Insect & Nematode Collection,
School of Earth & Environmental Sciences,
Waite Campus,
University of Adelaide,
Glen Osmond SA 5064,
AUSTRALIA
Correspondence regarding this page should be directed to Andrew R. Deans at and John T. Jennings at
Page copyright © 2004 Andrew R. Deans and John T. Jennings
- First online 04 October 2004
Citing this page:
Deans, Andrew R. and Jennings, John T. 2004. Evanioidea. Version 04 October 2004 (complete). http://tolweb.org/Evanioidea/11170/2004.10.04 in The Tree of Life Web Project, http://tolweb.org/