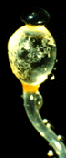Fungi
Eumycota: mushrooms, sac fungi, yeast, molds, rusts, smuts, etc.
Meredith Blackwell, Rytas Vilgalys, and John W. Taylor


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxPhylogeny modified from Bruns et al. 1991, 1993
Introduction
The organisms of the fungal lineage include mushrooms, rusts, smuts, puffballs, truffles, morels, molds, and yeasts, as well as many less well-known organisms (Alexopoulos et al., 1996). About 70,000 species of fungi have been described; however, some estimates of total numbers suggest that 1.5 million species may exist (Hawksworth, 1991; Hawksworth et al., 1995).
As the sister group of animals and part of the eukaryotic crown group that radiated about a billion years ago, the fungi constitute an independent group equal in rank to that of plants and animals. They share with animals the ability to export hydrolytic enzymes that break down biopolymers, which can be absorbed for nutrition. Rather than requiring a stomach to accomplish digestion, fungi live in their own food supply and simply grow into new food as the local environment becomes nutrient depleted.
Most biologists have seen dense filamentous fungal colonies growing on rich nutrient agar plates, but in nature the filaments can be much longer and the colonies less dense. When one of the filaments contacts a food supply, the entire colony mobilizes and reallocates resources to exploit the new food. Should all food become depleted, sporulation is triggered. Although the fungal filaments and spores are microscopic, the colony can be very large with individuals of some species rivaling the mass of the largest animals or plants.
 image info
image info
Figure 1: Hyphae of a wood-decaying fungus found growing on the underside of a fallen log. The metabolically active hyphae have secreted droplets on their surfaces. Copyright ? M. Blackwell 1996.
Prior to mating in sexual reproduction, individual fungi communicate with other individuals chemically via pheromones. In every phylum at least one pheromone has been characterized, and they range from sesquiterpines and derivatives of the carotenoid pathway in chytridiomycetes and zygomycetes to oligopeptides in ascomycetes and basidiomycetes.
Within their varied natural habitats fungi usually are the primary decomposer organisms present. Many species are free-living saprobes (users of carbon fixed by other organisms) in woody substrates, soils, leaf litter, dead animals, and animal exudates. The large cavities eaten out of living trees by wood-decaying fungi provide nest holes for a variety of animals, and extinction of the ivory billed woodpecker was due in large part to loss, through human activity, of nesting trees in bottom land hardwoods. In some low nitrogen environments several independent groups of fungi have adaptations such as nooses and sticky knobs with which to trap and degrade nematodes and other small animals. A number of references on fungal ecology are available (Carroll and Wicklow, 1992; Cooke and Whipps, 1993; Dix and Webster, 1995).
However, many other fungi are biotrophs, and in this role a number of successful groups form symbiotic associations with plants (including algae), animals (especially arthropods), and prokaryotes. Examples are lichens, mycorrhizae, and leaf and stem endophytes. Although lichens may seem infrequent in polluted cities, they can form the dominant vegetation in nordic environments, and there is a better than 80% chance that any plant you find is mycorrhizal. Leaf and stem endophytes are a more recent discovery, and some of these fungi can protect the plants they inhabit from herbivory and even influence flowering and other aspects of plant reproductive biology. Fungi are our most important plant pathogens, and include rusts, smuts, and many ascomycetes such as the agents of Dutch elm disease and chestnut blight. Among the other well known associations are fungal parasites of animals. Humans, for example, may succumb to diseases caused by Pneumocystis (a type of pneumonia that affects individuals with supressed immune systems), Coccidioides (valley fever), Ajellomyces (blastomycosis and histoplasmosis), and Cryptococcus (cryptococcosis) (Kwon-Chung and Bennett, 1992).
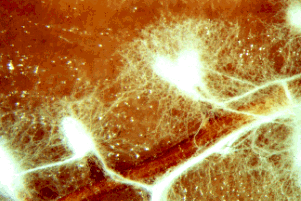 image info
image info
Figure 2: The fluffy white hyphae of the mycorrhizal fungus Rhizopogon rubescens has enveloped the smaller roots of a Virginia pine seedling. Note that some of the mycelium extends out into the surrounding environment. Copyright ? J. B. Anderson 1996.
 image info
image info
Figure 3: Entomophthora, "destroyer of insects", is the agent of a fungual infection that kills flies. After their death the fungal growth erupts through the fly cuticle, and dispersal by forcible spore discharge is a source of inoculum for infection of new flies. Copyright ? G. L. Barron 1996.
Fungal spores may be actively or passively released for dispersal by several effective methods. The air we breathe is filled with spores of species that are air dispersed. These usually are species that produce large numbers of spores, and examples include many species pathogenic on agricultural crops and trees. Other species are adapted for dispersal within or on the surfaces of animals (particularly arthropods). Some fungi are rain splash or flowing water dispersed. In a few cases the forcible release of spores is sufficient to serve as the dispersal method as well. The function of some spores is not primarily for dispersal, but to allow the organisms to survive as resistant cells during periods when the conditions of the environment are not conducive to growth.
Fungi are vital for their ecosystem functions, some of which we have reviewed in the previous paragraphs. In addition a number of fungi are used in the processing and flavoring of foods (baker's and brewer's yeasts, Penicillia in cheese-making) and in production of antibiotics and organic acids. Other fungi produce secondary metabolites such as aflatoxins that may be potent toxins and carcinogens in food of birds, fish, humans, and other mammals.
A few species are studied as model organisms that can be used to gain knowledge of basic processes such as genetics, physiology, biochemistry, and molecular biology with results that are applicable to many organisms (Taylor et al., 1993). Some of the fungi that have been intensively studied in this way include Saccharomyces cereviseae, Neurospora crassa, and Ustilago maydis.
Most phyla appear to be terrestrial in origin, although all major groups have invaded marine and freshwater habitats. An exception to this generality is the flagellum-bearing phylum Chytridiomycota, which probably had an aquatic origin; however, some chytrid species do occur in terrestrial environments, primarily as plant pathogenic fungi.
Characteristics
Fungi are characterized by non-motile bodies (thalli) constructed of apically elongating walled filaments (hyphae), a life cycle with sexual and asexual reproduction, usually from a common thallus, haploid thalli resulting from zygotic meiosis, and heterotrophic nutrition. Spindle pole bodies, not centrioles, usually are associated with the nuclear envelope during cell division. The characteristic wall components are chitin (beta-1,4-linked homopolymers of N-acetylglucosamine in microcrystalline state) and glucans primarily alpha-glucans (alpha-1,3- and alpha-1,6- linkages) (Griffin, 1994).
 image info
image info
Figure 4: Portion of a hypha of a zygomycete stained with a blue dye to show the many nuclei present. Many other fungi have septations that devide the hyphae into compartments that usually contain one to several nuclei per compartment. Copyright ? M. Blackwell 1996.
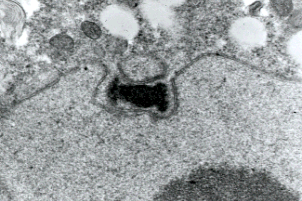 image info
image info
Figure 5: Transmission electron micrograph showing duplicated spindle pole body of a prophase I meiotic nucleus of a basidiomycete Exobasidium. Only chytrids among fungi have centrioles and lack spindle pole bodies. Copyright ? Beth Richardson 1996.
Exceptions to this characterization of fungi are well known, and include the following: Most species of Chytridiomycota have cells with a single, smooth, posteriorly inserted flagellum at some stage in the life cycle, and centrioles are associated with nuclear division. The life cycles of most Chytridiomycota are poorly studied, but some (Blastocladiales) are known to have zygotic meiosis (therefore, alternation between haploid and diploid generations). Certain members of Zygomycota, Ascomycota, and Basidiomycota may lack hyphal growth during part or all of their life cycles, and, instead, produce budding yeast cells. Most fungal species with yeast growth forms contain only minute amounts of chitin in the walls of the yeast cells. A few species of Ascomycota (Ophiostomataceae) have cellulose in their walls, and certain members of Chytridiomycota (Coelomomycetales) lack walls (Alexopoulos et al., 1996).
Fossil Record
Based on the available fossil record, fungi are presumed to have been present in Late Proterozoic (900-570 mya). Terrestrial forms of purported ascomycetes are reported in associations with microarthropods in the Silurian Period (438-408 mya) (Sherwood-Pike and Gray, 1985). Fossil hyphae in association with wood decay and fossil chytrids and Glomales-Endogenales representatives associated with plants of the Rhynie Chert are reported from the Devonian Period (408-360 mya) (Hass et al., 1994; Remy et al., 1994a, 1994b; Taylor et al., 1994a, 1995b). Fungal fossil diversity increased throughout the Paleozoic Era (Taylor et al., 1994b) with all modern classes reported in the Pennsylvanian Epoch (320-286 mya).
A first attempt to match molecular data on fungal phylogeny to the geological record shows general agreement, but does point out some conflicts between the two types of data (Berbee and Taylor 1993).
Biogeography
Wherever adequate moisture, temperature, and organic substrates are available, fungi are present. Although we normally think of fungi as growing in warm, moist forests, many species occur in habitats that are cold, periodically arid, or otherwise seemingly inhospitable. It is important to recognize that optimum conditions for growth and reproduction vary widely with fungal species. Diversity of most groups of fungi tends to increase in tropical regions, but detailed studies are only in their infancy (Isaac et al., 1993).
Although many saprobic and plant pathogenic species with low substrate specificity and effective dispersal systems have broad distributions, gene flow appears to be restricted in many fungi. For these species large bodies of water such as the Atlantic and Pacific Oceans create barriers to gene exchange. Some distributions are limited by substrate availability, and dramatic examples come from parasites of Gondowanan plants; one of these is the Southern Hemisphere distribution of the ascomycete Cyttaria, corresponding with part of the distribution of its host plant Nothofagus. The fossil record shows that fungi were present in Antarctica, as is the case for other organisms with Gondwanan distributions. Arthropod associates also may show distributions throughout part or all of a host range, and some fungal species (ex. wood wasp associates) occur outside the range of the associated arthropod.
Notable Fungi
- The largest basidiocarp known is that of a Rigidioporus ulmarius
in a
shady, hidden-away corner of the Royal Botanic Gardens, Kew, Surrey, England.
This basidiocarp is mentioned in the Guinness Book of Records (Matthews, 1994). At the
beginning of each new year the Annual Mensuration Ceremony of the
basidiocarp takes place. On 19 January 1996 the basidiocarp had increased to
170 cm maximum length (up from 159 in 1995) and 146 cm maximum width (up
from 140 in 1995). It also grew 4 cm taller from the soil level, now
measuring 54 cm. The weight of the basidiocarp has been estimated to be 284 kg (625 pounds)! Other large basidiocarps are those of
a puffball almost 9
feet in circumference in Canada (over 48 pounds) and a basidiocarp of the
sulfur mushroom in England (100 pounds).
 image info
image info
Figure 6: Largest basidiocarp world record holder Rigidioporus ulmarius at Kew. The basidiocarp is shown in its largest dimension (170 cm or over 5 1/2 feet). Copyright ? D. Pegler 1996.
- Reproductive structures clearly can be very large, but what about the body of the fungus, which often is hidden from view within the substrate? One fungus body constructed of tubular filaments (hyphae) was brought to our attention when molecular techniques were used to show that it was extensive (37 acres and an estimated blue whale equivalent size of 110 tons). The Michigan fungus clone (Armillaria bulbosa) grew in tree roots and soil. This report drew attention to an even larger fungal clone of Armillaria ostoyae, reported earlier in the state of Washington, which covered over 1,500 acres. Each clone began from the germination of a single spore over a thousand years ago. Although they probably have fragmented and are no longer continuous bodies, such organisms give us cause to think about what constitutes an individual.
- Penicillium chrysogenum is known for its production of the antibiotic penicillin. Although other antibiotics are produced by a variety of organisms, penicillin was the first to be developed. In the spring of 1996 a long dried out culture of the original isolate prepared by its discoverer, Sir Alexander Fleming in the late 1920s, was auctioned by Sotheby's of London and sold to a pharmaceutical company for 23 000 pounds. This price is insignificant when one considers the worth of this fungus, not only in sales of penicillin, but in terms of illnesses cured and lives saved. In the past a simple scratch sometimes could produce a fatal infection such as the one that resulted in the death of Tad Lincoln, the son of a U. S. president. However, misuse of penicillin and other antibiotics has resulted in selection of resistant microorganisms, and the threat of untreatable bacterial infections and diseases (for example, tuberculosis and syphilis) has returned.
- Fungal spores fill the air we breathe. On many days in some localities the number of fungal spores in the air far exceeds the pollen grains. Fungal spores also cause allergies; however, unlike seasonal pollen production, some fungi can produce spores all year long. The largest number of fungal spores ever sampled was over 5.5 million per cubic foot in Wales (Matthews, 1994).
- Basidiomycetes have always attracted a lot of attention because some of them have large basidiocarps, but the realization that all fungi are important in ecosystem function has drawn more attention to microscopic forms as well. For example a report on the secret sex life of a yeast-like ascomycete human pathogen, Coccidioides immitis, made a headline of the New York Times (6 February 1996, p. B7). This fungus causes Valley Fever and is endemic in parts of the southwestern United States. Although no one has been able to observe sexual reproduction in this species, molecular studies show genetic diversity that is best explained by occurrence of sexual reproduction in the life cycle.
- Another yeast-like ascomycete reported in the Dallas Morning News (28 August 1995, p. 8D) lives in the gut of cigar beetles and is essential to the beetle's health. Without the gut fungi to detoxify the plant material of toxins, the beetles would be poisoned. Keep on the lookout for other reports of fascinating fungal feats.
Discussion of Phylogenetic Relationships
As presently delimited, the kingdom Fungi is believed to constitute a monophyletic group that shares some characters with animals such as chitinous structures, storage of glycogen, and mitochondrial UGA coding for tryptophan. Only species of the basal Chytridiomycota possess the primitive character of a single smooth, posteriorly inserted flagellum (Barr, 1992; Cavalier-Smith, 1987, 1995).
The branch uniting the fungi and animals is well-supported from nuclear small subunit ribosomal RNA gene (SSU rDNA, or 18S rDNA) sequence analysis, and also has been supported with studies of elongation factor and three other proteins: alpha- and beta-tubulin and actin (Baldauf and Palmer, 1993; Bruns et al., 1991; Wainright et al., 1993). However, the hypothesis has been challenged by comparison of RNA polymerase gene sequences and reanalysis of SSU rDNA data (Rodrigo et al., 1994; Sidow and Thomas, 1994).
In the absence of many derived morphological (other than hyphal growth in some species) and biochemical characters, Chytridiomycota (previously characterized by the presence of a flagellate cell in the life cycle) now has been redefined by SSU rDNA sequence analysis. The study also has shown that some non-flagellate species traditionally placed in Zygomycota actually are chytrids (ex. Basidiobolus ranarum) (Nagahama et al., 1995).
Zygomycota as traditionally delimited on the basis of often non-septate hyphae and production of zygospores is not monophyletic. The position of some lineages such as that of Glomales and of Engodonales-Mortierellales is unclear, but they too may lie outside Zygomycota as independent lineages basal to the Ascomycota-Basidiomycota lineage (Bruns et al., 1993; K. O'Donnell, personal communication, 1996).
Evidence from shared morphological characters such as regularly septate hyphae and a dikaryotic stage (two separate and different nuclei in a single hyphal segment) in the life cycle, usually has been interpreted as support for a close relationship between Basidiomycota and Ascomycota. SSU rDNA adds support for this hypothesis (Berbee and Taylor, 1992).
Molecular characters have been essential for phylogenetic analysis in cases when morphological characters are convergent, reduced, or missing among the taxa considered. This is especially true of species that never reproduce sexually, because characters of sexual reproduction traditionally have been the basis for classification of Fungi. Use of molecular characters allows asexual fungi to be placed among their closest relatives. Excluded groups previously considered to be Fungi are stramenopiles (Oomycota, Hyphochytriomycota, and Labyrinthulomycota) and slime molds (Myxomycota, Plasmodiomycota, Dictyosteliomycota, Acrasiomycota) (Bhattacharya et al., 1992; Leipe et al., 1994; Van der Auwera et al., 1995).
References
Alexopoulos, C. J., C. W. Mims, and M. Blackwell. 1996. Introductory Mycology (4th Ed.). John Wiley and Sons, New York, USA. 868p.
Baldauf, S. L., and J. D. Palmer. 1993. Animals and fungi are each other's closest relatives: congruent evidence form multiple proteins. Proceedings of the National Academy of Sciences (USA) 90:11558-11562.
Barr, D. J. S. 1992. Evolution and kingdoms of organisms from the perspective of a mycologist. Mycologia 84:1-11.
Berbee, M. L., and J. W. Taylor. 1992. Two ascomycete classes based on fruiting-body characters and ribosomal DNA sequence. Molecular Biology and Evolution 9:278-284.
Berbee, M. L., and J. W. Taylor. 1993. Dating the evolutionary radiations of the true fungi. Canadian Journal of Botany 71:1114-1127.
Bhattacharya, D., L. Medlin, P. O. Wainright, E. V. Ariztia, C. Bibeau, S. K. Stickel, and M. L. Sogin. 1992. Algae containing chlorophylls a +c are paraphyletic: molecular evolutionary analysis of the Chromophyta. Evolution 46:801-1817.
Bruns, T. D., T. J. White, and J. W. Taylor. 1991. Fungal molecular systematics. Annual Review of Ecology and Systematics 22:525-564.
Bruns, T. D., R. Vilgalys, S. M. Barns, D. Gonzalez, D. S. Hibbett, D. J. Lane, L. Simon, S. Stickel, T. M. Szaro, W. G. Weisburg, and M. L. Sogin. 1993. Evolutionary relationships within the fungi: analysis of nuclear small subunit rRNA sequences. Molecular Phylogenetics and Evolution 1:231 241.
Carroll, G.C., and D.T. Wicklow. 1992. The Fungal Community: Its Organization and Role in the Ecosystem. Marcel Deker, Inc., New York.
Cavalier-Smith, T. 1987. The origin of Fungi and Pseudofungi. Pp. 339-353. In: Evolutionary Biology of the Fungi. Eds. A. D. M. Rayner, C. M. Brasier, and D. Moore. Cambridge University Press, Cambridge, United Kingdom.
Cooke, R.C., and J.M. Whipps. 1993. Ecophysiology of Fungi. Blackwell Scientific Pub., London, U.K.
Dix, N.J., and J.W. Webster. 1995. Fungal Ecology. Capman and Hall. London, U.K.
Griffin, D. 1993. Fungal Physiology (2nd Ed.). Wiley-Liss. New York.
Hasegawa, M., T. Hashimoto, J. Adachi, N. Iwabe, and T. Miyata. 1993. Early branchings in the evolution of eukaryotes: ancient divergence of Entamoeba that lacks mitochondria revealed by protein sequence data. Journal of Molecular Evolution 36:380-388.
Hass, H., T. N. Taylor, and W. Remy. 1994. Fungi from the Lower Devonian Rhynie Chert - mycoparasitism. American Journal of Botany 81:29-37.
Hawksworth, D. L. 1991. The fungal dimension of biodiversity: magnitude, significance, and conservation. Mycological Research 95:641-655.
Hawksworth, D. L., P. M. Kirk, B. C. Sutton, and D. N. Pegler. 1995. Ainsworth and Bisby's Dictionary of the Fungi (8th Ed.). CAB International, Wallingford, United Kingdom. 616p.
Isaac, S., J. C. Frankland, R. Watling, and A. J. S. Whalley. 1993. Aspects of Tropical Mycology. Cambridge University Press, Cambridge, U.K.
Kwon-Chung, K.J., and J.E. Bennett. 1992. Medical Mycology. Lea and Febiger, Philadelphia.
Leipe, D. D., P. O. Wainright, J. H. Gunderson, D. Porter, D. J. Patterson, F. Valois, S. Himmerich, and M. L. Sogin. 1994. The straminopiles from a molecular perspective: 16S-like rRNA sequences from Labyrinthula minuta and Cafeteria roenbergensis. Phycologia 33:369-377.
Nagahama, T., H. Sato, M. Shimazu, and J. Sugiyama. 1995. Phylogenetic divergence of the entomophthoralean fungi: evidence from nuclear 18S ribosomal RNA gene sequences. Mycologia 87:203-209.
Matthews, P. (Ed.). 1994. Guinness Book of Records. Bantum Books, New York. 819p.
Remy, W., T. N. Taylor, and H. Hass. 1994a. Early Devonian fungi - a blastocladalean fungus with sexual reproduction. American Journal of Botany 81:690-702.
Remy, W., T. N. Taylor, H. Hass, and H. Kerp. 1994b. Four hundred-million-year-old vesicular arbuscular mycorrhizae. Proceedings of the National Academy of Sciences (USA) 91:11841-11843.
Rodrigo, A. G., P. R. Bergquist, and P. I. Bergquist. 1994. Inadequate support for an evolutionary link between the metazoa and the fungi. Systematic Biology 43:578-584.
Sherwood-Pike, M. A., and J. Gray. 1985. Silurian fungal remains: probable records of the class Ascomycota. Lethaia 18:1-20.
Sidow, A., and W. K. Thomas. 1994. A molecular evolutionary framework for eukaryotic model organisms. Current Biology 4:596-603.
Taylor, J. W., B. Bowman, M. L. Berbee, and T. J. White. 1993. Fungal model organisms: phylogenetics of Saccharomyces, Aspergillus and Neurospora.. Systematic Biology 42:440-457.
Taylor, T. N., W. Remy, H. Hass 1994a. Allomyces in the Devonian. Nature 367:601-601.
Taylor, T. N., J. Galtier, B. J. Axsmith. 1994b. Fungi from the Lower Carboniferous of central France. Review of Palaeobotany and Palynology 83:253-260.
Taylor, T. N., W. Remy, H. Hass, H. Kerp. 1995a. Fossil arbuscular mycorrhizae from the Early Devonian. Mycologia 87:560-573.
Van der Auwera, G., R. De Baere, Y. Van de Peer, P. De Rijk, I. Van den Broeck, and R. De Wachter. 1995. The phylogeny of Hyphochytriomycota as deduced from ribosomal RNA sequences of Hyphochytrium catenoides. Molecular Biology and Evolution 12:671-678.
Wainright, P. O., G. Hinkle, M. L. Sogin, and S. K. Stickel. 1993. Monophyletic origins of the Metazoa: an evolutionary link with fungi. Science 260:340-342.
Information on the Internet
Popular Sites
- MykoWeb. WWW pages devoted to the science of mycology and the hobby of mushrooming.
- Introduction to the Fungi. UCMP Berkeley.
- Mycological Society of San Francisco. North America's largest local amateur mycological association.
- The Fungal Jungal. To further educate people about fungi, edible and otherwise, To encourage sustainable and responsible mushroom harvest, and preserve mushroom habitat.
- Tom Volk's Fungi.
- Dave Fischer's North American Mushroom Basics.
- MushroomExpert.Com.
- Forest Fungi.
- Pilze, Pilze, Pilze. In deutsch.
- Westf?lischen Pilzbriefe. In deutsch.
- Micologi Associati. Nell'italiano.
Directories, Databases & Collections
- The WWW Virtual Library: Mycology. A well indexed entrance to almost all mycology and fungal biology resources on the Internet.
- Mycology.Net. An internet site containing information about diversity of fungi.
- Mycorrhiza Information Exchange.
- Mycology Online. A WWW resource of clinically significant mycological information.
- Yahoo Mycology.
- Mycologists Online. World-wide directory for mycology and lichenology.
- Fungal Databases. Systematic Botany and Mycology Laboratory. Agricultural Research Service. United States Department of Agriculture. Beltsville, Maryland, USA.
- The Fifth Kingdom online. A mycological encyclopedia.
- Mycological and Lichenological Collection Catalogs. UCMP Berkeley.
- University of Alberta Microfungus Collection & Herbarium (UAMH).
- University of Michigan Fungus Collection.
- Mycological Herbarium. The Natural History Museums and Botanical Garden, University of Oslo.
- Herbarium Mycologicum. National Botanic Garden of Belgium.
- Index Fungorum. Names of fungi.
- Centraalbureau voor Schimmelcultures (CBS). Fungal Biodiversity Center - Utrecht, The Netherlands.
- Fungal Genetics Stock Center.
- Canadian Collection of Fungal Cultures.
Images
- Treasures from the Kingdom of Fungi. Taylor Lockwood's mushroom photography.
- Fungus plates painted under the supervision of Elias Fries. Department of Cryptogamic Botany, SwedishMuseumof NaturalHistory.
- Fungi Images on the Net.
- Fungi, Moulds and Lichens. BioImages: The Virtual Field-Guide (UK).
- Kinok-ya: Portraits of Mushrooms from Japan.
- George Barron's Website on Mushrooms and other Fungi.
- Eileen's Mushroom Mania.
- Herrera's Microfungi Home Page.
- Nathan's Fungi Page.
- Pamela's Mushrooms.
Research Labs & Projects
- Deep Hypha. NSF-funded Research Coordination Network (RCN) that is focused on developing robust phylogenetic hypotheses for the deep branches within Kingdom Fungi and enhanced research and educational tools in fungal systematics.
- AFTOL: Assembling the Fungal Tree of Life. Collaborative research in fungal phylogenetics.
- Systematic Botany and Mycology Laboratory. Agricultural Research Service. United States Department of Agriculture. Beltsville, Maryland, USA.
- Forest Mycology and Mycorrhiza Research Team. Forestry Sciences Laboratory, Corvallis, OR, USA.
- Cornell Center for Fungal Biology (CCFB).
- IUCN SSC Fungi Specialist Group Website.
- Bruns Lab. University of California at Berkeley. Ecology and evolution of fungi.
- Spatafora Lab. Oregon State University. Systematics and evolutionary biology of fungi.
- Taylor Lab. University of California at Berkeley. Evolutionary relationships of fungi, concentrating on the fungi that cause human disease.
- Thorn Lab. University of Western Ontario. Fungal ecology and systematics.
- Vilgalys Lab. Duke University. Natural history of fungi, including all aspects of their evolutionary biology, population genetics, and systematics.
- MycoSite. University of Oslo, Norway.
- University of Georgia Mycology.
- University of Tennessee Mycology Lab.
- Fungal Mitochondrial Genome Project (FMGP). B. Franz Lang, Universit? de Montr?al.
- Fungimap Australia. A collaborative project between professional and amateur mycologists and naturalists to gather information about the distribution of fungi throughout Australia.
- The Fungi of New Zealand. Manaaki Whenua Landcare Research.
- MycoKey. Thomas L?ss?e & Jens H. Petersen, University of Aarhus provide:
- Mapping of Macromycetes in Norway. A co-operation between the Norwegian Mycological Society and the Norwegian Ethno-Botanical Society with their local associations and the Museums of Natural History in Oslo, Bergen, Trondheim and Troms?.
- Comparative Studies on the Macrofungi of China and Eastern North America. Qiuxin Wu & Gregory M. Mueller, The Field Museum, Chicago.
- Survey of Northern Illinois and Indiana Fungi. John F. Murphy & Gregory M. Mueller. The Field Museum, Chicago.
- Macrofungi of Costa Rica. Roy E. Halling & Gregory M. Mueller.
- The Fungi of Kenya.
- Malawi Fungi.
- Moulds. Isolation, Cultivation, Identification. David Malloch, Department of Botany, University of Toronto.
Professional Societies
- The International Mycological Association. A group that represents mycologists and fungal biologists throughout the world.
- British Mycological Society.
- Mycological Society of America.
- Asociaci?n Latinoamericana de Micolog?a.
- Australasian Mycological Society.
- The International Society for Mushroom Science. To further the cultivation of edible (including medicinal) macrofungi.
Title Illustrations
| Scientific Name | Chytridium (Chytridiomycota) |
|---|---|
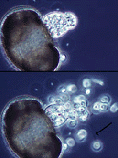
| Comments | Individual growing on a single pine pollen grain. Successive photos show zoospore release from the sporangium, and the arrow points to a flagellum. |
| Copyright | © 1996 H. Whisler, M. Fuller |
| Scientific Name | Pilobolus (Zygomycota) |
|---|---|
| Comments | Black sporangium atop swollen sporangiophore. Shortly, the swollen subsporangial vesicle will burst to send the sporangium flying. Herbivores eat the sporangium, and the enclosed mitospores germinate in the dung. The bright yellow carotenoid pigment enables the sporangium to orient to light (phototropism). If you look closely, you can see masses of nematodes on the vesicle; probably herbivore pathogens hoping to hitch a ride. |
| Specimen Condition | Live Specimen |
| Copyright | © 1996 Meredith Blackwell |
| Scientific Name | Laetiporus sulphureus (Basidiomycota) |
|---|---|
| Comments | Massed fruiting bodies of the chicken-of-the-woods. The tiny tubular filaments (hyphae) that make the body of this fungus (mycelium) are growing in the old, dead wood of a large cherry tree. Laetiporus is not a parasite, but the decay may weaken the tree so much that wind or ice storms can topple it. |
| Specimen Condition | Live Specimen |
| Copyright | © 1996 Meredith Blackwell |
| Scientific Name | Scarcoscypha coccinea (Ascomycota) |
|---|---|
| Comments | Fruiting body of the scarlet cup fungus. Hundreds of millions of meiospores (ascospores) are discharged from this cup, usually in puffs that produce visible clouds of spores. |
| Specimen Condition | Live Specimen |
| Copyright | © 1996 Joey Spatafora |
About This Page
Many thanks to Soren Rosendahl and Atul Batra for scanning photos and David Maddison and Atul Batra for page design advice.
Meredith Blackwell
Department of Plant Biology
Louisiana State University
Baton Rouge
Louisiana 70803, USA
Rytas Vilgalys
Department of Botany
Duke University
Durham, NC 27708-0338
USA
John W. Taylor
Department of Plant and Microbial Biology
321 Koshland Hall
University of California
Berkeley, CA 94720-3120
USA
Correspondence regarding this page should be directed to Meredith Blackwell at
Page copyright © 1996 Meredith Blackwell, Rytas Vilgalys, and John W. Taylor
- Content changed 14 February 2005
Citing this page:
Blackwell, Meredith, Vilgalys, Rytas, and Taylor, John W. 2005. Fungi. Eumycota: mushrooms, sac fungi, yeast, molds, rusts, smuts, etc.. Version 14 February 2005 (under construction). http://tolweb.org/Fungi/2377/2005.02.14 in The Tree of Life Web Project, http://tolweb.org/