Paleognathae
David P. Mindell and Joseph W. Brown

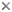
This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxReferences
Amadon, D. 1947. An estimated weight of the largest known bird. Condor 49: 159-164.
Ansari, H., N. Kakagi, and M. Sasaki. 1988. Morphological differentiation of sex chromosomes in three species of ratite birds. Cytogenetics and Cell Genetics 47: 185-188.
Baccetti, B., A. G. Burrini, and E. Falchetti. 1991. Spermatozoa and relationships in palaeognath birds. Biology of the Cell 71: 209-216.
de Boer, L. E. M. 1980. Do the chromosomes of the kiwi provide evidence for a monophyletic origin of the ratites? Nature 287: 84-85.
Caspers, G. J., J. Wattel, and W. W. de Jong. 1994. Alpha A-crystallin sequences group tinamou with ratites. Molecular Biology and Evolution 11: 711-713.
Cooper, A., C. Lalueza-Fox, S. Anderson, A. Rambaut, J. Austin, and R. Ward. 2001. Complete mitochondrial genome sequences of two extinct moas clarify ratite evolution. Nature 409: 704-707.
Cooper, A., C. Mourer-Chauvir?, G. K. Chambers, A. von Haeseler, A. C. Wilson, and S. P??bo. 1992. Independent origins of New Zealand moas and kiwis. Proceedings of the national Academy of Sciences of the USA 89: 8741-8744.
Cracraft, J. 1974. Phylogeny and evolution of the ratite birds. Ibis 116: 494-521.
Cubo, J. 2003. Evidence for speciational change in the evolution of ratites (Aves: Palaeognathae). Biological Journal of the Linnean Society 80: 99-106.
Davies, S. J. J. F. 1967. Sexual dimorphism in the emu. Emu 67: 23-26.
Davies, S. J. J. F. 2002. Ratites and Tinamous. Oxford University Press, New York.
Edwards, S. V., B. Fertil, A. Giron, and P. J. Deschavanne. 2002. A genomic schism in birds revealed by phylogenetic analysis of DNA strings. Systematic Biology 51: 599-613.
Freitag, S., and T. J. Robinson. 1993. Phylogeographic patterns in mitochondrial DNA of the ostrich (Struthio camelus). Auk 110: 614-622.
Garc?a-Moreno, J., and D. P. Mindell. 2000. Rooting a phylogeny with homologous genes on opposite sex chromosomes (gametologs): a case study using avian CHD. Molecular Biology and Evolution 17: 1826-1832.
Garc?a-Moreno, J., M. D. Sorenson, and D. P. Mindell. 2003. Congruent avian phylogenies inferred from mitochondrial and nuclear DNA sequences. Journal of Molecular Evolution 57: 27-37.
H?rlid, A., A. Janke, and U. Arnason. 1997. The mtDNA sequence of the ostrich and the divergence between Paleognathous and Neognathous birds. Molecular Biology and Evolution 14: 754-761.
Harrath, O., and A. J. Baker. 2001. Complete mitochondrial genome sequences of extinct birds: ratite phylogenetics and the vicariance biogeography hypothesis. Proceedings of the Royal Society of London Biological Sciences 268: 939-945.
Harrison, G. L., McLenachan, M. J. Phillips, K. E. Slack, A. Cooper, and D. Penny. 2004. Four new avian mitochondrial genomes help get to basic evolutionary questions in the late Cretaceous. Molecular Biology and Evolution 21: 974-983.
Huyen, L., C. D. Millar, and D. M. Lambert. 2002. A DNA test to sex ratite birds. Molecular Ecology 11: 851-856.. Nature 425: 175-178.
Lee, K., J. Feinstein, and J. Cracraft. 1997. The phylogeny of ratite birds: resolving conflicts between molecular and morphological data sets. Pp 173-208 in D. P. Mindell (ed.), Avian Molecular Evolution and Systematics. Academic Press, San Diego.
Mayr, G., and J. Clarke. 2003. The deep divergences of neornithine birds: a phylogenetic analysis of morphological characters. Cladistics 19: 527-553.
Osuga, D. T., and R. E. Feeney. 1968. Biochemistry of egg-white proteins of ratite group. Archives of Biochemistry and Biophysics 124: 560-574.
Parkse, K. C., and G. A. Clark Jr. 1966. An additional character linking ratites and tinamous, and an interpretation of their monophyly. Condor 68: 459-471.
Prager, E. M., A. C. Wilson, D. T. Osuga, and R. E. Feeney. 1976. Evolution of flightless land birds on southern continents: Transferrin comparison shows monophyletic origin of ratites. Journal of Molecular Evolution 8: 283-94.
Sibley, C. G., and C. Frelin. 1972. Egg-white protein evidence for ratite affinities. Ibis 114: 377-387.
Silynroberts, H., and R. M. Sharp. 1985. Preferred orientation if calcite in the ratite and tinamou eggshells. Journal of Zoology 205: 39-52.
Silynroberts, H., and R. M. Sharp. 1986. Preferred orientation if calcite in the Aepyornis eggshell. Journal of Zoology 208: 475-478.
Stapel, S. O., J. A. Leunissen, M. Versteeg, J. Wattel, and W. W. de Jong. 1984. Ratites as oldest offshoot of avian stem - evidence from alpha-crystallin A sequences. Nature 311: 257-259.
Tang, B., Y. H. Huang, L. Lin, X. X. Hu, J. Dong, P. Yao, L. Zhang, and N. Li. 2003. Isolation and characterization of 70 novel microsatellite markers from ostrich (Struthio camelus) genome. Genome 46: 833-840.
van Tuinen, M. C. G. Sibley, and S. B. Hedges. 1998. Phylogeny and biogeography of ratite birds inferred from DNA sequences of the mitochondrial ribosomal genes. Molecular Biology and Evolution 15: 370-376.
van Tuinen, M. C. G. Sibley, and S. B. Hedges. 2000. The early history of modern birds inferred from DNA sequences of nuclear and mitochondrial ribosomal genes. Molecular Biology and Evolution 17: 451-457.
Weir, K. A., and C. A. Lunam. 2004. A histological study of emu (Dromaius novaehollandiae) skin. Journal of Zoology 264: 259-266.
Zelenitsky, D. K., and S. P. Modesto. 2003. New information on the eggshell of ratites (Aves) and its phylogenetic implications. Canadian Journal of Zoology 81: 962-970.
Title Illustrations
| Scientific Name | Struthio camelus ssp. massaicus |
|---|---|
| Location | Masai Mara Game Reserve (Kenya) |
| Comments | Ostrich |
| Creator | Gerald and Buff Corsi |
| Acknowledgements | courtesy CalPhotos |
| Specimen Condition | Live Specimen |
| Copyright | © 2002 |
| Scientific Name | Dromaius |
|---|---|
| Location | Captive at Colin Mackenzie Sanctuary, Healesville, South Australia |
| Comments | Emus |
| Creator | Dr. G Dallas and Margaret Hanna |
| Acknowledgements | courtesy CalPhotos |
| Specimen Condition | Live Specimen |
| Copyright | © 2004 |
About This Page
David P. Mindell
Museum of Zoology and Department of Ecology and Evolutionary Biology
University of Michigan
Ann Arbor, MI 48109-1079
Joseph W. Brown
Museum of Zoology and Department of Ecology and Evolutionary Biology
University of Michigan
Ann Arbor, MI 48109-1079
Page copyright © 2004 David P. Mindell and Joseph W. Brown
- First online 14 December 2005
Citing this page:
Mindell, David P. and Brown, Joseph W. 2005. Paleognathae. Version 14 December 2005 (temporary). http://tolweb.org/Paleognathae/15837/2005.12.14 in The Tree of Life Web Project, http://tolweb.org/