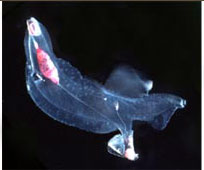
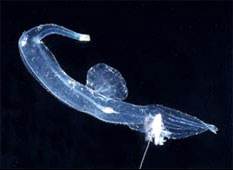
Carinarioidea
Heteropoda , heteropods, sea elephants
Roger R. Seapy- Atlantidae Rang, 1829
- Pterotracheidae
- Firoloida desmaresti
- Pterotrachea
- Pterotrachea coronata
- Pterotrachea hippocampus
- Pterotrachea scutata
- Carinariidae
- Carinaria
- Carinaria challengeri
- Carinaria cithara
- Carinaria cristata
- Carinaria galea
- Carinaria japonica
- Carinaria lamarcki
- Cardiapoda
- Cardiapoda placenta
- Cardiapoda richardi
- Pterosoma planum
Introduction
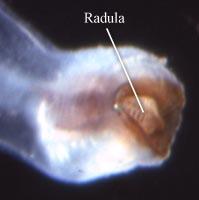
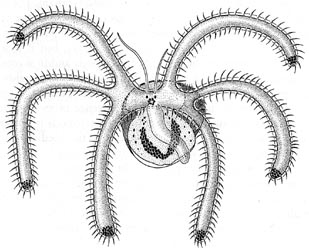
The heteropods are a group of pelagic snails (Class Gastropoda) that are found in moderate to low abundances, primarily in tropical to subtropical latitudes. Among the gastropods, they have three striking adaptations to the open ocean environment. The bodies and shells are largely transparent; only the buccal mass, eyes and viscera are opaque. The foot, which in bottom-dwelling snails is the sole-like structure used for crawling along the bottom, primarily forms a laterally-compressed swimming fin. The animal swims "upside-down" with the fin directed upward. The eyes are large and image-forming. They are active predators that visually locate their prey and capture them by protruding their radula (having elongate, sickle-shaped teeth) from the mouth at the tip of the proboscis. The presence øf a mobile, trunk-like proboscis is responsible for their common name, "sea elephants."
Diagnosis
Gastropod molluscs with:- Paired image-forming eyes with large, spherical lenses.
- Laterally-flattened ventral swimming fin.
Characteristics
- Eyes
- Eye forward-looking, with large, spherical lens.
- Eye with basal, ribbon-like retina.
- Eye shape, viewed from dorsal aspect, generally tubular to triangular.
- Wall of eye partially (unpigmented region forms a "window") to completely pigmented.
- Eye enclosed in gelatinous capsule.
- Foot
- Foot laterally compressed into swimming fin.
- Foot (swimming fin) usually with posteroventral sucker.
- Foot in atlantids forms anterior swimming fin and posterior opercular lobe with operculum (see Title figure).
- Proboscis and radula
- Proboscis a muscular, mobile extension of the head.
- Proboscis bears terminal buccal mass with radula.
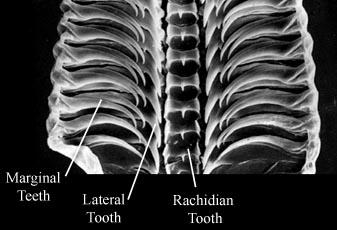
- Radula taenioglossate (tooth formula = 2-1-R-1-2).
- Lateral and marginal radular teeth with elongated shafts and hook-shaped ends.
- Shell
- Shell present (Atlantidae, Carinariidae) or absent (Pterotracheidae).
- Shell calcareous (aragonitic) and/or composed of cartilaginous-like material (conchiolin).
- Shell with prominent keel.
Figure. Dorsal view, left eye of Pterotrachea hippocampus, small animal (about 20 mm body length), Hawaiian waters. Eye with triangular shape. Photograph copyright ? 2005 R. Seapy.
Figure. Right side view, swimming fin and sucker of Carinaria japonica, male. copyright ? 2005 R. Seapy.
Figure. Dorsal view of head and proboscis (left) and buccal mass with radula (right) of Pterotrachea scutata, female, Hawaiian waters. Copyright ? 2005 R. Seapy.
Figure. Dorsal views of an atlantid radula. Left - Photomicrograph using dissection microscope. Right - Photomicrograph using scanning electron microscope. Photographs copyright ? 2005 R. Seapy (left) and G. Richter (right).
Figure. Left - Side view of Atlanta peroni with transparent shell, Hawaiian waters. Keel surrounds the outermost shell whorl. Right - Side view of transparent shell of Carinaria galea, Hawaiian waters. Large, flaring keel extends to the right in the photograph. Shell surrounds visceral mass and gills, all of which is attached to the body by a thick stalk. Photographs copyright ? 2005 R. Seapy.
Comments
The three families can be distinguished by the following characters:
| Family | Adult shell | Body can retract into shell | Adult body size | Location of swimming fin |
|---|---|---|---|---|
| Atlantidae | present | yes | microscopic (< ca. 1 cm) | opposite visceral mass |
| Carinariidae | present | no | macroscopic (> ca. 2 cm) | opposite visceral mass |
| Pterotracheidae | absent | n/a | macroscopic (> ca. 2 cm) | between visceral mass and head |
Life History
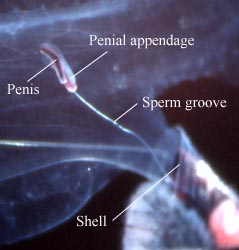
Heteropods are dioecious (separate sexes) and exhibit sexual dimorphism. Males have a prominent penis and penial appendage (function unknown) and the fin sucker in pterotracheids is present only in males. Also, there are sexual differences in radular morphology in some atlantids.
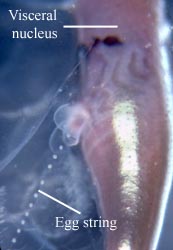
Male heteropods package sperm into spermatophores. Sperm are transferred to the penis by an external ciliated groove where they are packed into spermatophores. Spermatophores are transferred to females during copulation (mating behavior has only been observed in Pterotrachea hippocampus; Harbison in Lalli and Gilmer, 1989). Fertilized eggs are usually layed in mucoid egg strings which eventually break free from the female, except in Firoloida desmaresti which has a permanently attached tubular filament which holds the developing embryos (Owre, 1964).
Figure. Left - Side view of posterior region of trunk of Carinaria japonica, male, Hawaiian waters. Penis and penial appendage lie parallel to each other; penis contains brown spermatophore. Right - Side view of visceral mass (here, compacted into an elongate, brownish visceral nucleus) of Pterotrachea coronata, female, Hawaiian waters. The egg string is released from the oviduct opening, which is surrounded by transparent "lips". Photographs copyright ? 2005 R. Seapy.
All heteropods have a free-swimming, planktotrophic veliger larva. Larvae hatch from the egg within a few days of fertilization. The veliger possesses a dextrally (right-hand) coiled, calcareous shell and a flexible, chitinous operculum. The larval velum (ciliated swimming and feeding organ) is initially small and bilobed. With growth each lobe forms into two (pterotracheids) or three (atlantids and carinariids) long and slender lobes. Following metamorphosis the larval shell is retained as the protoconch of the adult shell (in atlantids and carinariids) or is cast off (in pterotracheids, which lack an adult shell). The larval operculum is either retained (atlantids) or cast off (carinariids and pterotracheids). The duration of the larval stage is not known, nor is age or life span of the adults. The largest heteropods are the carinariids (to 500 mm in Carinaria cristata) followed by the pterotracheids (to 330 mm in Pterotrachea coronata) (Lalli and Gilmer, 1989).
Figure. Left - Oral view of veliger larva of Carinaria lamarcki with six slender, ciliated velar lobes. Drawing from Thiriot-Quievr?ux (1973). Right - Late veliger larva of Oxygyrus keraudreni, shell diameter about 1.0 mm, from Hawaiian waters. Velar lobes retracted into shell, and head, proboscis and opercular lobe extend from shell. Photograph copyright ? 2005 R. Seapy.
Natural History
All heteropods swim with their ventral side directed upward and the dorsal side, with the shell (when present) and visceral mass, downward. Steady swimming is by undulations of the fin. In atlantids, a considerable side-to-side body motion is induced by the large fin, although the shell and keel act to partially offset the fin's movements. The large, elongate bodies of the carinariids and pterotracheids greatly dampen the side-to-side body motion. In the latter two groups, flexion of the trunk and tail are used in accelerated swimming during pursuit of prey or evasion of predators. The bodies of pterotracheids are the most elongate and streamlined of the three families, and they are presumably the fastest swimmers.
The atlantids are negatively buoyant. During the day they must swim to maintain position. At night, however, they secrete strands of buoyant mucus, up to 0.5 m long, from which they are suspended (Lalli and Gilmer, 1989; Newman, 1990). Carinariids and pterotracheids have been observed to float motionless without sinking (Lalli and Gilmer, 1989). Their neutral buoyancy results from the large amounts of gelatinous tissue, mostly in the trunk and tail, that is made positively-buoyant by ion regulation; replacement of heavier sulfate ions by lighter chloride ions (up to 75% in Pterotrachea coronata) (Denton and Shaw, 1961).
Heteropods are carnivores that feed during the day, locating their prey visually. The eyes are particularly unusual because they have a narrow, strip-like retina. Image formation is accomplished by scanning-eye movements. In atlantids, the only family studied to date, the retina is rotated through a 90° arc, from downward vertical to horizontal (Land, 1982). Presumably, prey are located beneath the animal by light reflected off the prey's body. In all heteropods, prey are captured by elongate, hooked radular teeth (described above). In the atlantids, the fin sucker is large and is used to hold the prey while pieces of tissue are torn off and ingested. In the carinariids and pterotracheids prey are ingested whole. Because the fin sucker is much smaller in these two latter groups, it is directed ventrally to posteroventrally, and it is spatially separated from the mouth by the long trunk; its primary function is presumably to hold males and females together during mating.
Heteropods feed on a variety of zooplanktonic prey. Atlantids feed preferentially on other gastropods, especially shelled pteropods (Richter, 1968; Newman, 1990), and carinariids feed selectively on soft bodied prey (e.g., salps, doliolids and chaetognaths in Carinaria japonica; Seapy, 1980). Little is known of the feeding in pterotracheids. Defense against predators is based largely on transparency although the buccal mass, pigmented region of the eyes, and the visceral mass (or visceral nucleus) are opaque. Pterotrachea uses reflectivity and behavioral orientation to reduce the visibility of these structures to upward-searching predators (Seapy and Young, 1986).
References
Denton, E. J. and T. I. P. Shaw. 1961. The buoyancy of gelatinous marine animals. J. Physiol., London 161: 14P-15P (Proceedings).
Lalli, C. M. and R. W. Gilmer. 1989. Pelagic snails. The biology of holoplanktonic gastropod snails. Stanford: Stanford University Press. 259 pp.
Land, M. F. 1982. Scanning eye movements in a heteropod mollusc. J. Exp. Biol. 96: 427-430.
Newman, L. 1990. The taxonomy, distribution and biology of Atlanta gaudichaudi Souleyet, 1852 (Gastropoda, Heteropoda) from the Great Barrier Reef. Amer. Malacol. Bull. 8: 85-94.
Newman, L. 1998. Superfamily Carinarioidea, pp. 804-808. In: P. L. Beesley, G. L. B. Ross and A. Wells (eds), Mollusca: the southern synthesis. The Fauna of Australia. Vol. 5, Pt. B. Melbourne: CSIRO Publ.
Owre, H. B. 1964. Observations on development of the heteropod molluscs Pterotrachea hippocampus and Firoloida desmaresti. Bull. Mar. Sci. Gulf Caribb. 14: 529-538.
Richter, G. 1968. Heteropoden und Heteropodenlarven im Oberfl?chenplankton des Golfs von Neapel. Pubbl. Staz. Zool. Napoli 36: 346-400.
Richter, G. 1974. Die Heteropoden der "Meteor"-Expedition in den Indischen Ozean, 1964/65. "Meteor" Forsch.-Ergebn. (D), 17: 55-78.
Richter, G. and R. R. Seapy. 1999. Heteropoda, pp. 621-647. In: D. Boltovskoy (ed.), South Atlantic Zooplankton. Leiden: Backhuys Publ.
Seapy, R. R. 1980. Predation by the epipelagic heteropod mollusk Carinaria cristata forma japonica. Mar. Biol. 60: 137-146.
Seapy, R. R., C. M. Lalli and F. E. Wells. 2003. Heteropoda from western Australian waters, pp.513-546. In: F. E. Wells, D. I. Walker and D. S. Jones (eds.), The marine flora and fauna of Dampier, Western Australia. Perth: Western Australia Museum.
Spoel, S. van der. 1976. Pseudothecosomata, Gymnosomata and Heteropoda (Gastropoda). Utrecht: Bohn, Scheltema & Holkema. 484 pp.
Spoel, S. van der, L. Newman and K. W. Estep. 1997. Pelagic molluscs of the world. World Biodiversity Data Base CD-ROM Series. Amsterdam: Expert Center for Taxonomic Identification (ETI).
Seapy, R. R. and R. E. Young. 1986. Concealment in epipelagic pterotracheid heteropods (Gastropoda) and cranchiid squids (Cephalopoda). J. Zool. Soc. London (A) 210: 137-147.
Tesch, J. J. 1949. Heteropoda. Dana Rept. No. 34. 53 pp.
Thiriot-Qui?vreux, C. 1973. Heteropoda. Oceanogr. Mar. Biol., Ann. Rev. 11: 237-261.
About This Page
Correspondence regarding this page should be directed to Roger R. Seapy at
Page copyright © 2005
- First online 15 February 2005
- Content changed 16 February 2005
Citing this page:
Seapy, Roger R. . 2005. Carinarioidea . Heteropoda , heteropods, sea elephants. Version 16 February 2005 (under construction). http://tolweb.org/Carinarioidea/27801/2005.02.16 in The Tree of Life Web Project, http://tolweb.org/