Clusiodes
Owen Lonsdale and Steve Marshall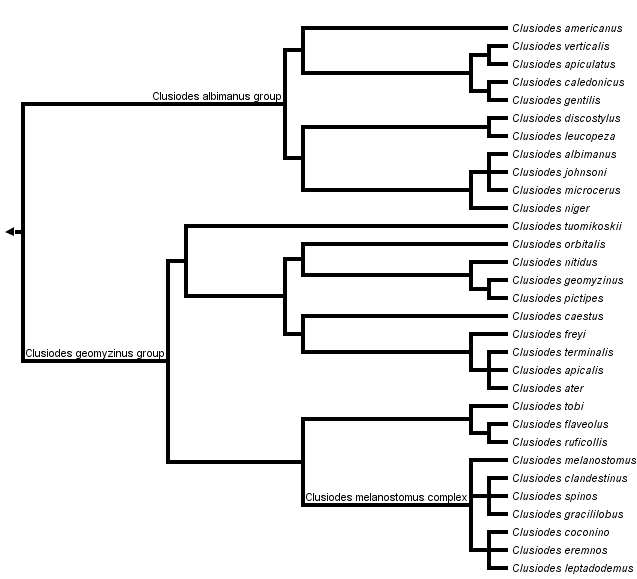

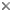
This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
Caloren & Marshall (1998) described the behaviour, biology and faunistics of the New World Clusiodes, and Rohácek (1995) described that for species in the Czech and Slovak Republics.
European species of Clusiodes and Hendelia beckeri have been reared from puparia collected in numerous species of rotting deciduous tree trunks including aspen, ash, birch, poplar and alder (Caloren & Marshall, 1998; Rohácek, 1995); several puparia and larvae of Clusiodes ruficollis have also been collected from under the bark of felled hornbeam in Britain (Withers, 1985). North American Clusiodes have been reared from dead tulip trees and likely develop in elm and maple (Caloren & Marshall, 1998). Clusiodes species overwinter as puparia or late-instar larvae in the larval habitat (Caloren & Marshall, 1998).
Adults of both genera are found predominantly on hardwood, but several Clusiodes species have been collected off of softwood trees such as pine, hemlock and fir (Caloren & Marshall, 1998;Rohácek, 1995; Withers, 1985). North American Clusiodes have also been found in association with rotting fungus (Caloren & Marshall, 1998). We have also found small dung baits, particularly those placed on fallen wood that is bare and exposed, to attract adults of Clusiodes and Hendelia in the New World. Sasakawa (1998) notes that Clusiodes and Sobarocephala have been collected with Malaise traps or fruit fly traps baited with putrid pork.
Fallen trunks are usually the most reliable microhabitat at which to collect adult Hendelia and Clusiodes, since they are often used as lekking sites. Rohácek (1995) noted that pale, conspicuous trunks (generally fallen trunks without bark) were selected by males of several Clusiodes species to attract females. Rohácek (1995) also observed numerous teneral specimens of C. ruficollis emerging from a moist, rotten beech trunk; these newly emerged adults were seen to search for new logs to use them as mating places; males patrolled these logs defending territory, but females visited the mating sites only briefly to copulate, then searched for similar, but more rotten logs to oviposit in. North American Clusiodes have been noted to prefer treefalls (both new and old) in sunspots, where males perform wing-flicking displays (alone or with conspecifics) and 'ambush-style attacks', in addition to the territorial patrolling and head-to-head confrontations previously noted in European species (Caloren & Marshall, 1998).
Characteristics
The north temperate Clusiodes, like its tropical and south temperate sister-genus Hendelia, can be characterized by reduced anterior and posterior fronto-orbitals, interfrontal bristles, well developed median genal bristles, swollen spermathecal ducts, a large subapical disc on the ventral receptacle, a longitudinal desclerotization on the female seventh sternite, a crested fin-like phallapodeme, a distiphallus that is rod-like basally with a membranous distal section, and an elongate pregonite that is perpendicular to the long axis of the hypandrium (Lonsdale & Marshall, in manuscript).

Figure. Top: External male terminalia of select European taxa, left lateral and posterior (left to right): Clusiodes pictipes (Zetterstedt); C. geomyzinus (Fallen); C. freyi Tuomikoski; C. apicalis (Zetterstedt); C. caledonicus (Collin); C. gentilis; C. verticalis (Collin). Bottom: C. apicalis (Zetterstedt), hypandrial complex, left lateral. © Owen Lonsdale.
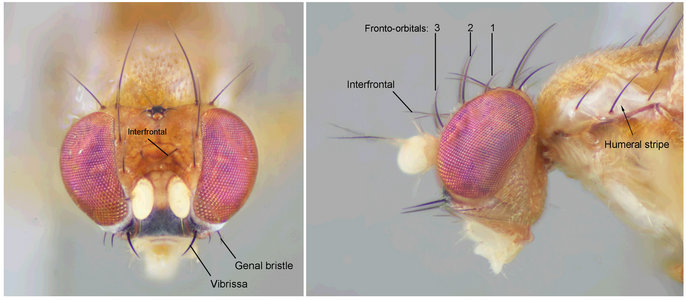
Head bristles of Clusiodes melanostomus.
Clusiodes can be separated from Hendelia by having a pubescent arista, evenly spaced fronto-orbitals, one pair of lateral white stripes on the scutum, and large parallel surstyli that sometimes have an anterodistal process (Lonsdale & Marshall, in manuscript). Clusiodes species are sometimes black in colouration, but a number of species have a yellow vittate scutum. Most Clusiodes, particularly the smaller ones, are generally matt, setulose and rounder in appearance than other Clusiidae. Species of Clusiodes also have shorter legs (usually thin and cylindrical) and heads that are generally rounded with a broadly curved frons, narrow face and blunt features.


Habitus of Clusiodes gracilolobus (top) & C. ater (bottom). © Owen Lonsdale
Discussion of Phylogenetic Relationships
Hendelia and Clusiodes are sister genera, supported by the synapomorphies listed above. They form a clade with Heteromeringia and Tranomeringia in the Clusiodinae based on telescoped and darkly pigmented spermathecae.
Clusiodes and Hendelia show a clear north/south division, with Clusiodes almost entirely north temperate and Hendelia almost entirely tropical and south temperate in distribution. Most Clusiodes clades include widely separated sister species that often occur on separate continents; this diverse aggregate of Nearctic and Palaearctic species support the theory that trans-oceanic speciation events have been frequent in the genus (Caloren & Marshall, 1998).
Clusiodes can be divided into two well-supported Holarctic clades, the C. albimanus and C. geomyzinus species groups, which largely overlap each other's distributions. The C. albimanus group is most obviously characterized by bicoloured fore tarsi, and the C. geomyzinus group is characterized by two well-developed dorsocentral bristles, a large bulging gena, short hypandrial bristles on a swollen basal lobe and a transverse sclerotized ridge on the female sixth tergite. A revision of the New World Clusiodes has been published by Caloren & Marshall (1998), and a World revision focusing on Old World species is currently being prepared (Lonsdale & Marshall, in manuscript).
References
Caloren, D.C. & S.A. Marshall. 1998. A revision of the New World species of Clusiodes Coquillett (Diptera: Clusiidae). Studia Dipterologica 5:261-321.
Rohacek, J. 1999. Clusiidae. Folia Fac. Sci. Nat. Univ. Masaryk. Brun., Biol. 100: 291-293.
Withers, P. 1985. Notes on some British Clusiidae and reduction of Clusiodes facialis (Coll.) to synonymy. Proceedings of the Transactions of the British Entomological Natural History Society 18: 63-64.
Sasakawa, M. 1998. Family Clusiidae. In. Contributions to a Manual of Palaearctic Diptera; Volume 3: Higher Brachycera. L. Papp & B. Darvas (eds.), pp. 219-225. Science Herald, Budapest.
Title Illustrations

| Scientific Name | Clusiodes ater Melander & Argo |
|---|---|
| Specimen Condition | Live Specimen |
| Life Cycle Stage | Adult |
| Copyright |
© Steve Marshall

|
| Scientific Name | Clusiodes apiculatus Malloch |
|---|---|
| Specimen Condition | Live Specimen |
| Sex | Female |
| Life Cycle Stage | Adult |
| Copyright |
© Steve Marshall

|
| Scientific Name | Clusiodes melanostomus complex |
|---|---|
| Specimen Condition | Live Specimen |
| Life Cycle Stage | Adult |
| Copyright |
© Steve Marshall

|
About This Page
Owen Lonsdale

University of Guelph, Canada
Steve Marshall

University of Guelph, Canada
Correspondence regarding this page should be directed to Owen Lonsdale at and Steve Marshall at
Page copyright © 2005 Owen Lonsdale and Steve Marshall
- First online 25 August 2005
- Content changed 20 August 2007
Citing this page:
Lonsdale, Owen and Marshall, Steve. 2007. Clusiodes. Version 20 August 2007 (under construction). http://tolweb.org/Clusiodes/27684/2007.08.20 in The Tree of Life Web Project, http://tolweb.org/









 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site